

Rob Devine, Business Development Manager, Life Sciences
Life sciences manufacturing is changing faster than traditional production models can keep up. Today’s facilities must deliver extreme precision, navigate tightening regulations, and respond to rising patient demand, all while managing workforce shortages and increasingly complex product portfolios.
Manufacturers are shifting from legacy systems to intelligent automation not because it’s optional, but because it has become critical to modern life sciences operations. Recent industry research reinforces a clear trend: resilient, compliant, and scalable automation systems are now essential for long-term competitiveness.
In this article
- Smart life sciences automation is becoming essential as manufacturers face workforce shortages, tighter regulatory requirements, rising costs, and pressure to accelerate time-to-market — making scalable automation a competitive necessity.
- Automation systems that embed precision technologies, track-and-trace capabilities, digital documentation, and compliance alignment with standards like GMP and 21 CFR Part 11 improve product quality while reducing audit risk and manual error.
- Intelligent manufacturing tools — including robotics, AI-driven workflows, digital twins, and modular architectures — enable faster scale-up, improved workforce efficiency, and greater flexibility for small-batch and complex product production.
- Organizations that invest in smart manufacturing and medical device automation can reduce operational strain, improve regulatory compliance, and build resilient facilities designed to adapt as product demand and technology changes.
The pressures reshaping life sciences manufacturing
The global life sciences sector faces intense pressure to innovate quickly while maintaining uncompromising safety and quality. At the same time, the demand for personalized medicine and advanced therapies is accelerating.
Manufacturers are now expected to produce smaller batches, maintain rigorous documentation, and ensure real-time quality control across increasingly complex operations. Automation helps bridge this gap by enabling scalable production with consistency, traceability, and compliance built into every step.
Recent industry insights show how deeply technology adoption is influencing the sector. Approximately 95% of life sciences manufacturers are using or evaluating smart manufacturing technologies to improve agility and resilience. Automation is no longer a “future investment.” For many manufacturers, it has become an urgent need.
Yet implementing automation — whether retrofitting existing assembly lines or integrating digital technologies — introduces its own challenges.
1. Workforce shortages that limit growth
The life sciences sector requires workers with rare combinations of engineering, regulatory, analytical, and operational expertise. Unfortunately, talent supply is not keeping pace with industry demand. The Global Life Science Risk Report 2024 found talent shortages to be one of the industry’s top emerging risks, with companies expanding into new markets and leaning heavily on automation simply to keep up.
2. Increasing regulatory complexity
Regulatory expectations — spanning GMP, GAMP 5®, ISO 9001, and 21 CFR Part 11—have expanded significantly in both scope and technical rigor. This creates significant operational challenges, with 42% of life sciences organizations identifying regulatory compliance as a top barrier to commercializing medical products.
3. Rising cost pressures and supply chain volatility
Modern life sciences manufacturers must produce more specialized products, more frequently, in smaller batches, and with tighter margins. Recent industry analyses highlight rising labor costs, expanding product diversity, unstable supply chains, and shrinking margins as key contributors to mounting operational strain across life sciences manufacturing.
4. Demand for faster time‑to‑market
Patients and providers expect faster delivery of diagnostics, devices, biologics, and therapeutics. Meanwhile, manufacturers face growing expectations to release higher‑quality products at higher volume — without extending timelines. Technology adoption is accelerating as companies race to close this gap and improve both speed and reliability, with many life sciences manufacturers already using or actively evaluating smart manufacturing technologies.
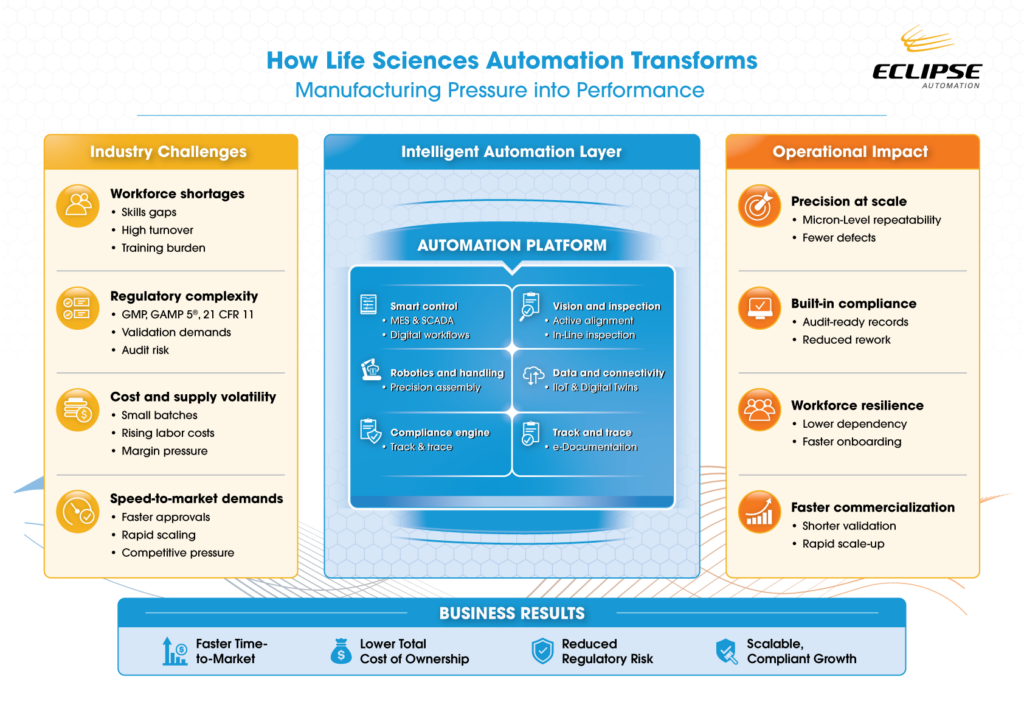
What automation solves for life sciences
Automation does far more than replace manual tasks; it embeds resilience, traceability, and quality into the core of manufacturing operations. It creates an integrated ecosystem combining data analytics, machine vision, smart controls, and digital monitoring to optimize every stage of production.
For every challenge manufacturers face in life science automation, there are solutions, each with reachable outcomes that do anything from improving efficiency and ROI to addressing internal cultural shifts. These are just a few examples:
| Precision and repeatability at scale | – Vision-based active alignment for micro-precision assembly – High-accuracy dispensing of reagents, epoxies, and lyophilized beads – Integrated laser welding and advanced plastic bonding technologies for durable seals – Continuous or asynchronous web processing for high-volume consumables Outcome: Faster throughput, fewer errors, and more consistent production across every unit |
| Improved quality and built-in compliance | – Embedded compliance directly into production workflows – Track-and-trace systems and automated documentation for audit-ready operations – MES or SCADA platforms aligned with 21 CFR Part 11 – ISO-aligned validation processes combined with in-line inspection Outcome: Consistent quality with reduced manual review and operational risk |
| Workforce optimization | – Collaborative robotics (“cobots”) and AI-driven systems to handle repetitive or hazardous tasks – Digital workflows, clear SOPs, and automated instructions to reduce errors – Supports onboarding, knowledge transfer, and workforce continuity – Allows teams to focus on quality improvement and innovation Outcome: Operational continuity despite labor shortages and skill gaps |
| Faster scale-up | – Smart factories, IIoT platforms, and modular automation architectures – Digital twins and simulation technologies for pre-deployment validation – Rapid adaptation to changing production needs or batch sizes Outcome: Reduced risk, faster commercialization, and accelerated innovation |
The future of the intelligent life sciences factory
Industry forecasts suggest the next generation of life sciences manufacturing will be defined by smart factories — connected environments where data, generative AI, and composable digital platforms drive decisions and automation adapts in real time.
Many organizations anticipate that nearly all processes that can be automated will be automated within the next five years. Life sciences manufacturers that embrace this shift now will be better prepared to address future demand and regulatory complexity, while delivering therapies, devices, and diagnostics that change lives.
With mounting workforce challenges, rising regulatory burdens, growing product complexity, and pressure to accelerate speed‑to‑market, manufacturers must build smarter, more flexible systems designed for modern realities. By embracing intelligent automation, manufacturers can improve precision, accelerate innovation, and maintain compliance in even the most demanding environments.
Market momentum already reflects this shift. Automation in the life sciences industry is a fundamental requirement for operational excellence. Eclipse stands ready to guide this transformation, helping manufacturers design and implement smarter, more flexible automation systems built for modern life sciences manufacturing realities.

Looking to accelerate innovation while maintaining compliance and quality? Book a discovery call to discuss life sciences automation solutions tailored to your manufacturing environment.
